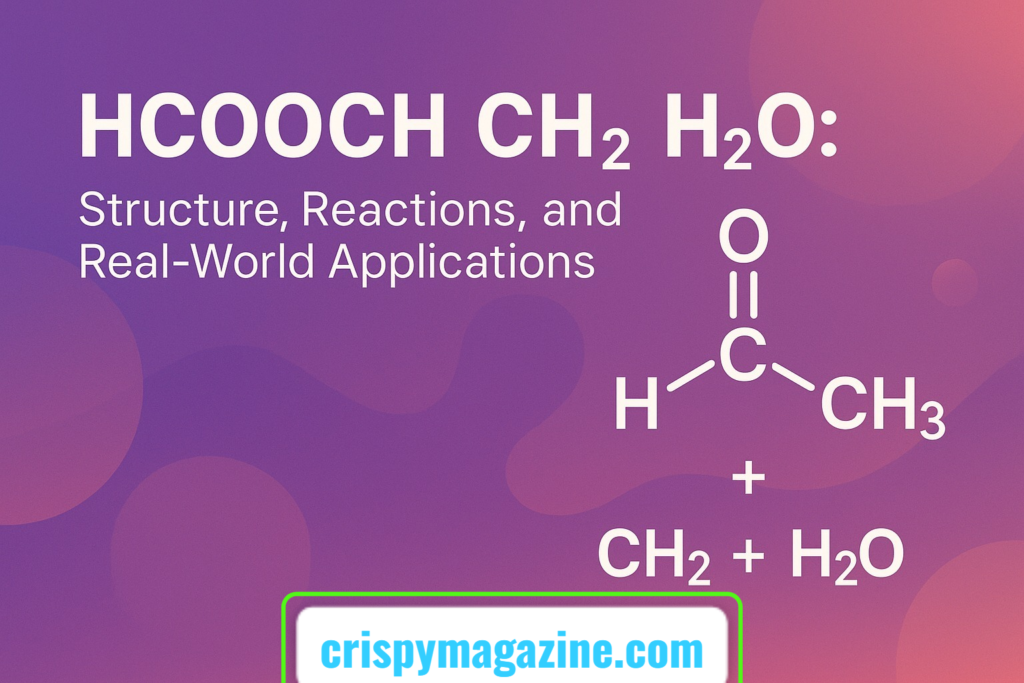
Introduction: Unveiling the Trio – HCOOCH, CH₂, and H₂O
In the vast landscape of organic chemistry, certain combinations of molecules stand out due to their unique interactions and versatile applications. One such intriguing ensemble is HCOOCH CH₂ H₂O. This trio comprises methyl formate (HCOOCH₃), a methylene group (CH₂), and water (H₂O). Each component brings distinct chemical properties to the table, and their interplay is pivotal in various chemical reactions and industrial processes.
Understanding the Components
Methyl Formate (HCOOCH₃)
Methyl formate is the simplest ester of formic acid. It is a colorless, volatile liquid with a pleasant odor, commonly used as a solvent and intermediate in organic synthesis. Its chemical structure features a formyl group (HCO-) bonded to a methoxy group (-OCH₃), making it susceptible to nucleophilic attacks, especially in hydrolysis reactions.
Methylene Group (CH₂)
The methylene group is a highly reactive moiety, often existing transiently in reactions as a carbene (:CH₂). It plays a crucial role in various organic transformations, including cyclopropanation and insertion reactions. In the context of HCOOCH CH₂ H₂O, the methylene group can act as a bridge or intermediate, facilitating the formation of complex molecules.
Water (H₂O)
Water, the universal solvent, is indispensable in chemical reactions. Its polar nature allows it to dissolve a wide range of substances, and it actively participates in hydrolysis and hydration reactions. In the HCOOCH CH₂ H₂O system, water not only serves as a medium but also as a reactant, driving the transformation of esters and alkenes.
HCOOCH CH₂ H₂O Overview Table
| Category | Details |
|---|---|
| Full Name | Likely refers to a hydrated or solvated ester compound (e.g., formyl vinyl ester hydrate) |
| Chemical Structure | Contains: – Formate group (HCOO−) – Vinyl group (CH=CH₂) – Water (H₂O, possibly hydrogen-bonded) |
| Key Reactions | – Hydrolysis of ester bond – Addition reactions at C=C – Polymerization (via CH=CH₂) |
| Physical State | Liquid (assumed, depending on hydration level and temperature) |
| Applications | – Organic synthesis intermediate – Polymer precursor – Flavor/fragrance industries |
Key Reactions Involving HCOOCH CH₂ H₂O
Ester Hydrolysis
One of the fundamental reactions involving this trio is the hydrolysis of methyl formate in the presence of water, yielding formic acid and methanol:
HCOOCH₃ + H₂O → HCOOH + CH₃OH
This reaction is acid or base-catalyzed and is significant in both laboratory and industrial settings, particularly in the production of formic acid and methanol.
Alkene Hydration
When the CH₂ component represents an alkene, such as ethylene (CH₂=CH₂), it can undergo hydration in the presence of water and an acid catalyst to form ethanol:
CH₂=CH₂ + H₂O → CH₃CH₂OH
This reaction is a cornerstone in the synthesis of alcohols from alkenes, highlighting the versatility of the CH₂ group in organic chemistry.
Industrial Applications
The HCOOCH CH₂ H₂O system finds applications across various industries:
- Solvent Production: Methyl formate serves as a solvent in manufacturing processes, including the production of adhesives and resins.
- Pharmaceuticals: The hydrolysis products, formic acid and methanol, are precursors in synthesizing various pharmaceuticals.
- Agrochemicals: Formic acid is used as a preservative and antibacterial agent in livestock feed.
- Textile Industry: Formic acid plays a role in dyeing and finishing textiles, improving color fastness and fabric quality.
Environmental and Safety Considerations
While the components of HCOOCH CH₂ H₂O are valuable in industrial applications, they also pose certain risks:
- Methyl Formate: Flammable and can cause respiratory irritation; proper ventilation and protective equipment are essential during handling.
- Formic Acid: Corrosive to skin and eyes; requires careful storage and handling procedures.
- Methanol: Toxic if ingested, inhaled, or absorbed through the skin; necessitates strict safety protocols.
Implementing appropriate safety measures and environmental controls is crucial to mitigate these risks.
Conclusion
The combination of HCOOCH, CH₂, and H₂O represents a fascinating intersection of organic chemistry principles and practical applications. From fundamental reactions like ester hydrolysis and alkene hydration to their roles in various industries, these components exemplify the dynamic nature of chemical interactions. Understanding their properties and behaviors not only enriches our knowledge of chemistry but also informs safer and more efficient industrial practices.
Frequently Asked Questions (FAQs)
Q1: What is the significance of the HCOOCH CH₂ H₂O system in organic chemistry?
A1: This system illustrates key reactions like ester hydrolysis and alkene hydration, which are fundamental in synthesizing various organic compounds.
Q2: How is methyl formate utilized in industries?
A2: Methyl formate is used as a solvent in manufacturing processes, including the production of adhesives, resins, and as an intermediate in pharmaceuticals.
Q3: What safety precautions are necessary when handling components of HCOOCH CH₂ H₂O?
A3: Proper ventilation, use of personal protective equipment, and adherence to safety protocols are essential to prevent exposure to toxic and flammable substances like methyl formate and methanol.



